(Caesarea, Israel, March 28, 2024) –- V-Wave today announced it will present late-breaking data from its RELIEVE-HF pivotal trial at the American College of Cardiology’s (ACC) Annual Scientific Session & Expo taking place April 6-8 in Atlanta. RELIEVE-HF is a randomized, double-blind, placebo-procedure-controlled, multicenter, multinational, pivotal trial of the V-Wave® Ventura® Interatrial Shunt in symptomatic patients with reduced (HFrEF) or preserved (HFpEF) left ventricular ejection fraction at high risk of heart failure (HF)-related events.
“We’re pleased to present the RELIEVE-HF data at this year’s ACC meeting, as we continue to advance this important technology with the goal of bringing a much-needed treatment option to patients with advanced heart failure, a leading cause of death and hospitalization in the U.S. and worldwide,” said Neal Eigler, M.D., CEO of V-Wave.
Summary of Presentation
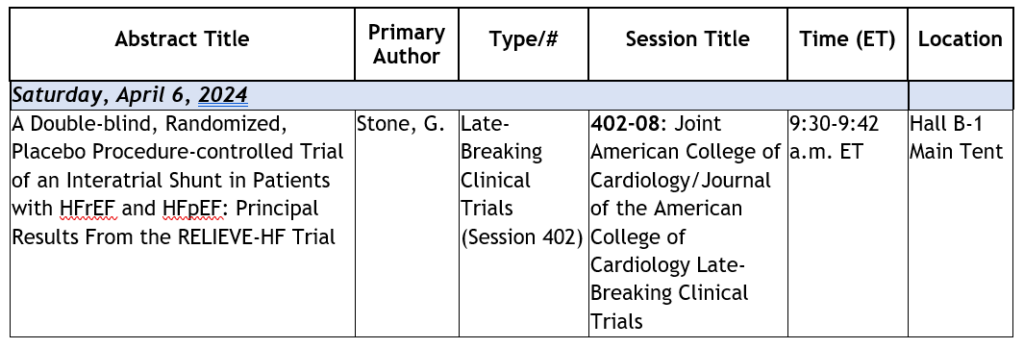
About RELIEVE-HF
The randomized, double-blind, placebo-procedure-controlled, multicenter, multinational, pivotal RELIEVE-HF clinical study (NCT03499236) evaluated the safety and effectiveness of the investigational V-Wave® Ventura® Interatrial Shunt for the treatment of HF. The study enrolled 508 patients with advanced HF on maximally tolerated guideline-directed medical therapy who were randomized 1:1 and stratified by LVEF to either the V-Wave® Ventura® Interatrial Shunt treatment arm or the control arm, who received a placebo procedure (sham). Additional patients were enrolled in an open-label (unblinded), roll-in arm, all of whom received the V-Wave® Ventura® Interatrial Shunt.
The primary effectiveness endpoint was a composite of death, heart transplant/left ventricular assist device (LVAD) implantation, HF hospitalization, outpatient HF worsening, and quality of life. The primary safety endpoint was measured as device-related and procedure-related major adverse cardiovascular or neurological device-related events (MACNE) at 30 days. Patients were followed for the primary analysis for a minimum of 12 months and a maximum of 24 months (median of 22 months). The study was performed under the U.S. Food and Drug Administration’s Breakthrough Devices Program and enrolled patients at 101 hospitals in 11 countries, including in the U.S., Canada, Israel, Australia and New Zealand.
About Heart Failure
Heart failure (HF) is a chronic, progressive disease in which the heart cannot pump enough blood to meet the body’s needs. HF affects 7 million people in the U.S, where it is a leading cause of death and hospitalization,[i] and 56.2 million people globally. When HF occurs, there is an increase in left atrial pressure, which causes fluid to back up in the lungs. This causes pulmonary congestion and makes it hard to breathe during activities or even at rest. In addition to shortness of breath, symptoms include general fatigue, tiredness, rapid weight gain due to fluid retention, and swollen legs and feet.
There are two types of HF. HFpEF is HF with preserved ejection fraction in which pressure builds up in the heart and lungs due to a stiffened left ventricle. This type of HF is largely associated with obesity and hypertension. In HFpEF, left ventricular ejection fraction (LVEF, a measure of the heart’s ability to pump oxygen-rich blood to the body) is >40. HFrEF is HF with reduced ejection fraction in which the left ventricle, the main pumping chamber of the heart, is weakened and usually enlarged and can’t effectively pump oxygen-rich blood to the body. In HFrEF, LVEF is <40.
Treatments for HF, which include medications, lifestyle changes and medical devices, typically focus on improving survival, reducing the risk of heart failure hospitalizations, and providing symptom relief. Despite advances in HF therapy, premature death, recurrent hospitalizations and deteriorating quality of life remain a large burden to patients, their families and the U.S. healthcare system.
About V-Wave
V-Wave is a privately held medical device company that was established in 2009 and is focused on developing innovative treatment options for people living with heart failure and cardiovascular disease. The company was built on a foundation of science, engineering and medicine and has decades of experience in these fields. V-Wave’s lead program is the investigational V-Wave® Ventura® Interatrial Shunt, which has Breakthrough Device Designation from the U.S. Food and Drug Administration and is designed to offer a safe, minimally invasive approach to treat heart failure. The company has offices in Israel and the U.S. For more information, please visit www.vwavemedical.com or V-Wave on LinkedIn.
Forward-Looking Statement
This press release contains certain forward-looking statements that involve risks and uncertainties, including statements related to clinical development and potential regulatory approval of V-Wave’s products. All forward-looking statements and other information included in this press release are based on information available to V-Wave as of the date hereof, and the Company assumes no obligation to update any such forward-looking statements or information. The company’s actual results could differ materially from those described in the company’s forward-looking statements.
CAUTION, the V-Wave® Ventura® Interatrial Shunt System is not available for sale in the United States or other countries. Limited by Federal (or United States) law to investigational use.
# # #
Contact
Elliot Fox, Real Chemistry
207-604-0956
# # #

Recent Comments